Use of vestibular sedatives for the treatment of peripheral vertigo in Colombia
Perfil de uso de sedantes vestibulares para el tratamiento de vértigo periférico en Colombia
Angela María Campos-Mahecha*, Luisa María Rojas-Herrera**.
* Otorhinolaryngology – Otology, Fundación Universitaria de Ciencias de la Salud (FUCS). ORCID: https://orcid.org/0000-0002-9653-4538
** Master's Degree in Clinical Pharmacology, Fundación Universitaria de Ciencias de la Salud (FUCS ORCID: https://orcid.org/0009-0007-2615-0663
How to cite: Campos-Mahecha AM, Rojas-Herrera LM. Perfil de uso de sedantes vestibulares para el tratamiento de vértigo periférico en Colombia. Acta otorrinolaringol. cir. cabeza cuello. 2025;53(1):pp-pp. Doi: 10.37076/acorl.v53i1.758
Article records:
Date of receipt: February 06, 2024
Date of revision: October 08, 2024
Date of acceptance: October 29, 2024
Corresponding author:
Dr. Luisa María Rojas Herrera
E-mail: lmrojas5@fucsalud.edu.co
Address: Calle 66 C # 61-01, apt 1101, torre 1; Bogotá, Colombia.
Mobile number: 3176181106
Abstract
Introduction: The inappropriate use of vestibular sedatives is associated with an in-creased risk of falls, hospitalizations, high costs, adverse events, and poorer clinical outcomes. Therefore, knowing medication consumption and its variation over time is crucial. The objective was to establish the usage profile of vestibular sedatives in the outpatient treatment of peripheral vertigo in patients over 18 years old insured under the Colombian Social Security System, from July 2021 to June 2022. Methods: A cross-sectional descriptive study used information from a medication dispensation database, evaluating sociodemographic, pharmacological, and clinical variables. Univariate and bivariate descriptive analysis were performed to establish a possible relationship between active ingredients, polypharmacy, interactions, synergy, and the prescribing doctor. Results: 22.776 patients were identified, with a median age of 56 years, where 64,65% were female. Concerning the utilization profile of vestibular sedatives: Dimenhydrinate, had the highest dispensation at 72,30%, indicated for pa-roxysmal vertigo and other peripheral vertigos, followed by Betahistine at 37,50%, Diphenhydramine at 6,50%, and Meclizine at 0,40%. Prolonged use of any vestibular sedative was observed in 7,0%, 74,45% exhibited polypharmacy, and the synergy summation was 4,80%. Conclusion: Dimenhydrinate had the highest prescription rate in Colombia. The significant percentage of polypharmacy and pharmacological management of benign paroxysmal vertigo is noteworthy. Therefore, individualized treatment is suggested, considering potential adverse reactions.
Keywords (MeSH): Vertigo; Vestibular Diseases; Pharmacoepidemiology; Drug Therapy; Betahistine; Dimenhydrinate; Meclizine; Diphenhydramine.
Resumen
Introducción: el uso inadecuado de sedantes vestibulares se asocia a mayor riesgo de caídas, hospitalizaciones, costos, eventos adversos y peores resultados clínicos. Por lo tanto, es importante tener conocimiento acerca del consumo de los medi-camentos y su variación en el tiempo. El objetivo fue establecer el perfil de uso de los sedantes vestibulares en el tratamiento ambulatorio de vértigo periférico en pacientes mayores de 18 años, asegurados al Sistema de Seguridad Social colombia-no, desde julio del 2021 hasta junio 2022. Métodos: estudio descriptivo transversal que utilizó información de una base de dispensación de medicamentos, donde se evaluaron variables sociodemográficas, farmacológicas y clínicas. Se realizó un aná-lisis descriptivo univariado y bivariado para establecer una posible relación entre los principios activos, polifarmacia, interacciones, sinergismo de suma y médico prescriptor. Resultados: se identificaron 22.776 pacientes, con una mediana de edad de 56 años, donde el 64,65% corresponde al sexo femenino. En cuanto al perfil de uso de sedantes vestibulares, el dimenhidrinato fue el sedante vestibular con mayor dispensación en un 72,30% para indicaciones como vértigo paroxístico benigno y otros vértigos periféricos, seguido de betahistina 37,50%, difenhidramina 6,50% y meclizina 0,40%. El uso prolongado de cualquier sedante vestibular se presentó en el 7,0%, el 74,45% presentó polifarmacia y el sinergismo de sumación fue del 4,80%. Conclusión: la mayor prescripción en Colombia fue para el dimenhidrinato. Llama la atención el alto porcentaje de polifarmacia y el manejo farmacológico del vértigo paroxístico benigno, por lo que se sugiere un tratamiento individualizado, valorando las posibles reacciones adversas.
Palabras clave (DeCS): vértigo, enfermedades vestibulares, farmacoepidemiología, tratamiento farmacológico, betahistina, dimenhidrinato, meclizina, difenhidramina.
Introduction
Vertigo is a common reason for consultation in the emergency department and outpatient services (1). Approximately 20%-40% of the general population is affected by this condition, and its incidence increases with age, reaching 39% in people over 80 years of age (2). Currently, the diagnostic approach to patients involves four categories that help classify patient symptoms: imbalance (a feeling of unsteadiness, lack of equilibrium, or abnormal gait), syncope (loss of consciousness resulting from cerebral hypoperfusion or cardiac arrhythmia), dizziness (a vague term correlated with a category of presyncope), and vertigo, defined as the sensation of an illusion of movement of the body and/or the environment when no actual movement is occurring. The most common symptoms reported are rotational dizziness and swaying, which are associated with nausea and emetic episodes. These concepts must be clearly understood, as they may overlap with each other (3).
Peripheral vertigo is the most common type of vertigo. It is usually benign and self-limited, which means that a large proportion of patients, despite having symptoms, do not seek medical care. As a result, these patients are not considered in statistics regarding this condition (4). In the United States, nearly 5.6 million physician visits per year are associated with vertigo, and a quarter of them are of peripheral or vestibular origin (1). The most common causes of vestibular vertigo are benign paroxysmal positional vertigo (BPPV), Ménière's disease, labyrinthitis, neuronitis, vestibular schwannoma, and vestibular migraine (1, 5, 6).
A study conducted in 2013 aimed to characterize the use of drugs that increase the risk of falls in patients suffering from dizziness who were treated at a neurotology service in Baltimore City, Maryland. It was found that 40% of patients were over 50 years of age, with a 7% greater probability of falls for each subsequent decade of age. It was also reported that, among patients over 65 years of age, 44% of men and 57% of women used multiple drugs: 39% of patients consumed psychotropic medications, 37% antihypertensives, and 8.2% opioids (7). This study concluded that patients using more than one type of medication had a higher risk of balance impairment, and it emphasized that benzodiazepine use tripled the probability of hospitalization due to a femoral fracture resulting from a fall (8, 9).
Vertigo has a significant impact on patients’ quality of life, since about 41% of them lose their jobs, 40% interrupt their activities of daily living, and 19% refrain from leaving their homes. Furthermore, they are at increased risk of falling, and the presence of vertigo is related to the development or exacerbation of psychiatric disorders. The impact of vertigo is even greater in people over 65 years of age, resulting in disability, alterations in physical functioning, social life, activities of daily living, and increased indirect health care costs, thus making it a relevant public health issue (4, 10-12). These patients need to be treated using a multidisciplinary approach that implements strategies to tailor treatments and safely reduce the amount of medications used by this population (7, 11, 13-15).
The objective of this study is to describe the most common medications used for the outpatient treatment of peripheral vertigo in patients over 18 years of age insured by the Colombian General System of Social Security in Health (SGSSS by its Spanish acronym) between July 2021 and June 2022.
Materials and methods
Cross-sectional study, carried out between July 1, 2021, and June 30, 2022, on the use of vestibular sedatives for outpatient treatment of peripheral vertigo in a population of approximately 9 million individuals registered in the SGSSS through four health promoting entities (EPS by its Spanish acronym). Data were gathered from a technological platform that collects information from the selected pharmaceutical provider.
The study population included adult individuals over 18 years of age, of either sex, who were prescribed vestibular sedatives (dimenhydrinate, betahistine, diphenhydramine, and meclizine) and were diagnosed according to the ICD-10 with Ménière's disease, BPPV, vestibular neuronitis, other peripheral vertigo, other vestibular disorders, vertiginous syndromes in diseases classified elsewhere, labyrinthitis, labyrinth fistula, labyrinth disorders, other specified inner ear disorders, and unspecified inner ear disorder.
Based on the information obtained by the logistics operator, a database was created in Excel 2020 (version 16.43), detailing socio-demographic variables (sex, age, insurance scheme, origin, prescribing physician's specialty, insurance company, and diagnosis) and pharmacological variables (name of the vestibular sedative, amount dispensed, pharmaceutical form, concentration, and estimated duration of dispensing). The number of defined daily doses (DDD) was also used as a variable to measure the use of the four active ingredients under study, following the recommendations of the World Health Organization (WHO) and expressed as DID (DDD per 1000 inhabitants per day) (16-18). Variables such as polypharmacy, possible risk of interactions, and additive synergism were also considered to identify them and make recommendations on the proper use of the medications.
Data analysis was performed using the Jamovi statistical software (version 2.2.5), which was employed to analyze the results obtained. A univariate analysis was conducted, presenting the results of qualitative variables as absolute and relative frequencies, which were subsequently organized in tables. Quantitative variables were presented as medians and interquartile ranges based on the results of the normality tests. In turn, the bivariate analysis included variables such as active ingredient and prescribing physician's specialty, active ingredient and polypharmacy, and active ingredient and interactions. The level of statistical significance was set at p <0.05.
This study was approved by the Ethics Committee for Research involving Human Subjects (CEISH for its Spanish acronym) of the Sociedad de Cirugía de Bogotá, Hospital de San José (FUCS for its Spanish acronym). As this is an observational study, it complies with Article 11 of Resolution 008430 dated October 4, 1993 (“Scientific, technical, and administrative standards for health research”) issued by the Colombian Ministry of Health, classifying it as a “Risk-free research” due to its retrospective design, in which no biological, physiological, psychological, or social variables of the individuals to be studied are modified.
Results
From July 1, 2021, to June 30, 2022, a total of 22 776 patients were treated in 32 departments of Colombia. The median age of the entire population was 56 years (interquartile range [IQR]: 42-68 years), with the highest number of prescriptions of vestibular sedatives found in the group of patients under 60 years of age (59.50%) and 64.65% (14 724/22 776) of prescriptions being for females, as shown in Figure 1.
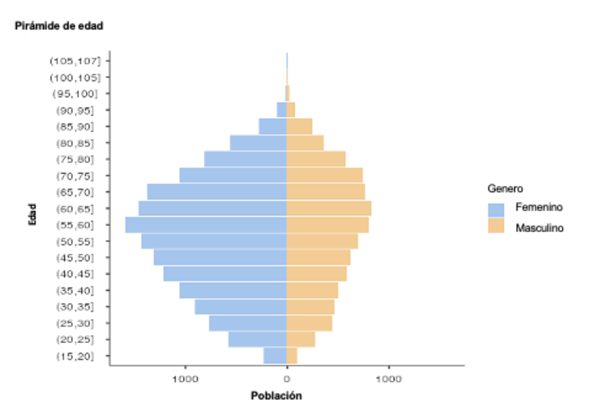
Age pyramid| Age| Population| Sex| Female | Male| 1000 | 0 | 1000 | (105,107) (100,105) (95,100) (90,95) (85,90) (80,85) (75,80)
Figure 1. Population pyramid showing the distribution by age and sex of users of vestibular sedatives for the treatment of peripheral vertigo in Colombia. Authors' own elaboration.
Concerning geographic regions, Bogotá had the highest number of prescriptions by active ingredient with 25.70% (5864/16,459) for dimenhydrinate and 8.30% (1891/8541) for betahistine. The second most frequent department was Valle del Cauca with 10.36% (2 355/22 776). A total of 81.60% of patients (18 586/22 776) were enrolled in the contributory insurance scheme and 18.40% (4190/22,776) in the subsidized insurance scheme.
Regarding the use of vestibular sedatives, the most prescribed active ingredient was dimenhydrinate in 72.30% (16 459/22 776), followed by betahistine in 37.50% (8 541/22 776), diphenhydramine in 6.50% (1 480/22 776), and meclizine in 0.40% (93/22 695), as shown in Figure 2. This group of drugs was mainly prescribed by general practitioners (n=22 672; 99.2%), internal medicine physicians (n=1 459; 6.40%), and otorhinolaryngologists (n=1 114; 4.9%).
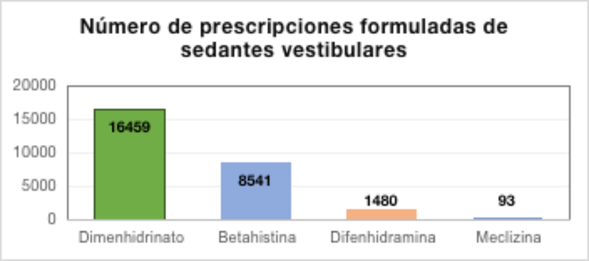
Number of vestibular sedative prescriptions | 20 000 | 15 000 | 10 000 | 5 000 | 0 | 16 459 | Dimenhydrinate | 8541 | Betahistine | 1480 | Diphenhydramine | 93 | Meclizine
Figure 2. Number of vestibular sedative prescriptions for the treatment of peripheral vertigo in Colombia. Authors' own elaboration.
The most frequent diagnoses associated with betahistine and dimenhydrinate were other peripheral vertigo in 65.30% (14 864/22 776) and benign paroxysmal vertigo in 35.70% (8 133/22 776). Labyrinth dysfunction diagnosis was observed in a percentage of less than 0.01% for both active ingredients. As for diphenhydramine and meclizine, the most frequent diagnosis was other peripheral vertigo in 4.40% (991/22,776) and 0.20% (52/22,776) respectively. There was no evidence of prescription of these two active ingredients under the diagnoses of labyrinth fistula, labyrinth dysfunction, and other specified disorders of the inner ear, as described in Table 1.
Table 1. Distribution of vestibular sedative prescription according to diagnosis for the treatment of peripheral vertigo in Colombia.
|
ICD-10. n (%) |
Betahistine n=8541 |
Dimenhydrinate n=16.459 |
Diphenhydramine n=1480 |
Meclizine n=93 |
|
Other peripheral vertigo
|
5 845 (25.70) |
10 695 (47.00) |
991 (4.40) |
52 (0.20) |
|
Benign paroxysmal vertigo
|
2 948 (12.90) |
6 076 (26.70) |
560 (2.50) |
35 (0.20) |
|
Vertiginous syndromes in diseases classified elsewhere
|
439 (1.90) |
679 (3.00) |
58 (0.30) |
16 (0.10) |
|
Ménière's disease
|
284 (1.20) |
104 (0.50) |
10 (0.0) |
1 (0) |
|
Other disorders of vestibular function |
74 (0.30) |
75 (0.37) |
10 (0.04) |
1 (0) |
|
Vestibular neuronitis
|
20 (0.18) |
29 (0.13) |
5 (0.07) |
0 (0) |
|
Labyrinthitis |
17 (0.18) |
38 (0.21) |
8 (0.02) |
7 (0) |
Authors' own elaboration.
As for the number of cumulative DDD during the study year, it was 600 542 for betahistine, 680 447 for dimenhydrinate, 54 970 for diphenhydramine, and 3 160 for meclizine. A calculation was made per 1 000 inhabitants per day (DID), where 0.21 DDD per day of dimenhydrinate, 0.18 DDD per day of betahistine, 0.02 DDD per day of diphenhydramine, and 0.001 DDD per day of meclizine were used for every 1 000 patients. The month with the highest usage of vestibular sedatives was June 2022, with a DDD number of 52 865.83, which represents the highest usage record for betahistine during the study year, as shown in Figure 3.
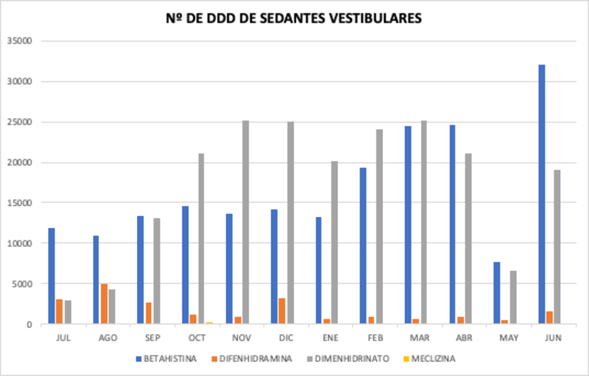
Number of DDD for vestibular sedatives
35 000 | 30 000 | 25 000 | 20 000 | 15 000 | 10 000 | 5 000 | 0 | JUL | AUG | SEP | OCT | NOV | DEC | JAN | FEB | MAR | APR | MAY | JUN | Betahistine | Diphenhydramine | Dimenhydrinate | Meclizine
Figure 3. Number of monthly DDD for vestibular sedatives in the treatment of peripheral vertigo in Colombia. Author's own elaboration.
The median dispensing time of vestibular sedatives was measured based on the number of months that the active ingredients evaluated were dispensed, showing that the majority received treatment for less than 3 months (90.70% for betahistine, 97.90% for dimenhydrinate, 97.90% for diphenhydramine, and 90.2% for meclizine). It was also noted that 0.2% (19/22 776) of patients receiving betahistine and 0.006% (1/22 776) receiving dimenhydrinate were continuously dispensed during the 12 months of the year under study. Two patients were continuously dispensed diphenhydramine and meclizine for a maximum of 9 and 7 months, respectively.
Overall, prolonged use of vestibular sedatives was found in 7.0% (1585/22,776) of all users of these drugs. In this study, prolonged use of vestibular sedatives was defined as use of the sedative for more than 3 months, and it was observed that the active ingredient with the longest use was betahistine in 4.0% (922/22,776), followed by dimenhydrinate in 2.10%, being higher in the group of patients older than 75 years of age.
Polypharmacy was detected in 74.45% (16 974/22 776) and additive synergism in 4.80% (1 094/22 776). Likewise, during the year under study, 4.7% (1 073/22 776) showed at least one additive synergism.
A higher frequency of interaction with antihypertensives was observed in 29.0% of patients who received dimenhydrinate (6 606/16 459) and 18.70% of those who received betahistine (4 254/8 541), with a similar behavior noted in the use of central nervous system depressants. Regarding dimenhydrinate, the most common interaction occurred with hyoscine bromide in 2.10% (471/22 776), while the remaining interactions evaluated occurred in a smaller proportion (0.5%).
The bivariate analysis showed that specialties such as otorhinolaryngology, neurology, and internal medicine prescribed less betahistine and dimenhydrinate, with a statistically significant difference with respect to other prescribers at a value of p <0.01. Polypharmacy was found in 54.40% of the dimenhydrinate group, mainly in the group of patients between 19-60 years of age, with a p<0,01. Similarly, dimenhydrinate was the most prescribed drug in this age group.
Discussion
The present study describes the prescription patterns of vestibular sedatives for peripheral vertigo in Colombia among patients enrolled in the SGSSS between July 2021 and June 2022. The results of this research are very useful to the medical and research community because they indirectly reveal the epidemiology of the disease and are a reference for pharmacovigilance systems.
Worldwide, vertigo is considered one of the most frequent reasons for seeking medical care, affecting approximately 15%-20% of the adult population (4, 19, 20-23, 28), with a higher incidence in the female sex and in patients over 60 years of age (24-27, 29). This is similar to what was observed in this study, in which 64.65% of the patients were female, although 59.50% of the population studied was in the range between 19 and 60 years of age. A German study from 2015 showed a decrease in the prevalence of the disease in older adults, in whom reduced vestibular function is attributed to geriatric syndrome, limited mobility, and to the fact that patients consider this imbalance as normal, which would result in fewer consultations and underdiagnosis of the condition (30).
This study focused on the use of vestibular sedatives for the treatment of peripheral vertigo in Colombia, where the most prescribed active ingredient was dimenhydrinate (72.30%), followed by betahistine (37.50%), diphenhydramine (6.50%), and meclizine (0.40%). To establish the usage pattern, a calculation was made per 1 000 inhabitants per day (DID), where 0.21 DDD per day of dimenhydrinate, 0.18 DDD per day of betahistine, 0.02 DDD per day of diphenhydramine, and 0.001 DDD per day of meclizine were used for each 1 000 patients. It is noteworthy that, despite being approved for vertigo treatment, the use of diphenhydramine and meclizine is low; however, these data cannot be compared with other countries, as there are no studies on the topic reported in the literature.
Balance depends on many factors, such as the peripheral vestibular system (semicircular canals, utricle, and saccule), the central nervous system, the vestibular nucleus, and the integration of visual and sensory information. This complex process involves neurotransmitters such as acetylcholine, glutamate, and glycine, which in turn are regulated by histamine, adrenaline, and norepinephrine (27, 28). Peripheral vertigo is treated both pharmacologically (during the acute phase or in times of crisis) and with vestibular rehabilitation (28, 31, 32).
In the present study, the most frequent diagnosis was other peripheral vertigo, found in 65.30% of the population studied. This is noteworthy because no precise diagnosis of the disease is established, resulting in a higher risk of interactions and polypharmacy. The second most frequent diagnosis was benign paroxysmal vertigo in 35.70%, which is consistent with the literature, as it is diagnosed in 17% to 42% of the population (28, 31-34).
Treatment for benign paroxysmal vertigo as recommended in the literature consists of repositioning maneuvers depending on the affected semicircular canal (35, 37, 41). In the present study, 35.70% of the population with this diagnosis received treatment, 26.70% received dimenhydrinate, 12.90% received betahistine and, to a lesser extent, diphenhydramine and meclizine. It should be noted that the literature advises against the use of vestibular suppressants for this condition, indicating that their administration should be limited to severely symptomatic patients, either before or after repositioning maneuvers, and for short periods of time, since prolonged pharmacotherapy interferes with central compensation and favors adverse reactions such as sedation, urinary retention, and falls (28). The drugs recommended for this group of patients are antihistamines (meclizine and diphenhydramine), benzodiazepines (diazepam and clonazepam), and promethazine (1, 33, 40). It should also be kept in mind that this condition is characterized by symptoms that can last from 6 to 20 days (37), with 20% of patients recovering spontaneously after one month of follow-up or up to 50% after three months (1, 38, 39, 41).
Vestibular neuritis is accompanied by high intensity autonomic symptoms, requiring the use of vestibular sedatives such as meclizine and dimenhydrinate (28, 42), which are the most commonly used in the United States. This contradicts the findings of this study, which revealed that betahistine (0.18%) was the most commonly used drug, followed by dimenhydrinate (0.13%), while meclizine was not recommended. A recent clinical trial demonstrated that treatment with dimenhydrinate and diazepam in the emergency department was equivalent (42, 43) but, given the adverse effects of benzodiazepines, antihistamines are preferred. (28). The literature recommends limiting the use of vestibular sedatives to the first few days after symptom onset (37).
The mechanism of action of betahistine is to modulate histamine neurotransmission and to be a partial antagonist of H1 receptor activity, with an increased antagonistic effect on histamine H3 receptors, resulting in increased histamine production and release (44, 45). Betahistine has also been proven to be involved in neuronal excitability and impulse generation in the lateral and medial vestibular nuclei (46). The H3 receptor plays an important role in vestibular compensation, behavioral restoration, and symptom relief (47, 48) as a result of its vascular effects on the cochlea; the effects of betahistine are maintained through the formation of the aminoethyl pyridine metabolite (48). A Cochrane review demonstrated that the use of betahistine may have a positive effect on the reduction of vertigo symptoms (49), but the level of evidence was low due to the low quality of the methods used.
For Ménière's disease, on the other hand, the prescription of vestibular sedatives was 1.40%, with betahistine being the most frequently prescribed in 1.20% of cases. Current scientific evidence recommends using vestibular sedatives during the acute phase or in crises (28). Multiple studies have been conducted to determine the effectiveness of betahistine for the intercritical phase of Ménière's disease, but results are still inconclusive. In a multicenter study, the use of betahistine was proposed as first-line treatment for the intercritical phase with doses ranging from 32 to 48 mg/day. In this study, it is recommended to prescribe betahistine for at least 3 months to patients who have had one to three seizures in the last 6 months (50). Moreover, a prospective, controlled clinical trial (BEMED) with betahistine doses of 48 mg/day and 48 mg three times a day failed to show a significant therapeutic effect compared to placebo (45). However, the latest Cochrane review on the efficacy of betahistine (51, 52) and two meta-analyses (53, 54) conclude that the use of betahistine is acceptable. A recent study reported that betahistine is rapidly metabolized by the intestinal and hepatic MAO-B enzyme, thus the combination of betahistine-selegiline (MAO-B inhibitor) reduces the dose of betahistine necessary to avoid new attacks (55).
This study assessed the median dispensation time of vestibular sedatives, finding that more drugs were dispensed in the first three months of treatment, which supports the acute use of these drugs. However, it is noteworthy that prolonged use is reported in a low percentage, despite the lack of evidence in the literature to support its use beyond the acute phase.
Considering the intensity of the symptoms and their impact on balance, vertigo is a disabling condition that affects normal daily activities and increases the risk of falls when it occurs in elderly patients. It is necessary to bear in mind that the population over 65 years of age exhibits changes in the pharmacokinetic and pharmacodynamic parameters of drugs and is prone to side effects related to changes in body composition and renal or hepatic failure, which can lead to greater vulnerability (56, 57).
The percentage of polypharmacy in this study was 74.45%. This finding suggests that the study population is at a high risk of polypharmacy, which is in turn associated with an increased risk of drug-drug interactions, falls, prolonged hospitalization, and even death. Therefore, educational programs should be implemented to modify the use of these drugs to reduce the incidence of side effects and to tailor treatment to each patient (58).
It is essential to rule out the possibility that ototoxic medications, including antihypertensives, diuretics, α-adrenergic receptor antagonists, or calcium channel blockers, are the cause for symptoms such as orthostatic hypotension or damage to the cochlear and vestibular receptors of the inner ear, leading to imbalance (29, 59). The possibility of suspending the causal drug should be evaluated in these patients (60). The present study identified a potential interaction between antihypertensives and dimenhydrinate in 29% of cases, and with betahistine in 18.70%, which should be assessed individually in each patient when prescribing these antihistamines.
The vestibular sedatives evaluated show several interactions inherent to their mechanism of action when acting as antihistamines. For example, in the present study, dimenhydrinate showed a possible interaction with hyoscine bromide in 2.10%, and both are anticholinergic drugs. Consequently, side effects such as tachycardia, ileus, urinary retention, and prolongation of the QT interval are exacerbated (56).
Diphenhydramine is a first-generation H1 receptor antihistamine, a metabolite of dimenhydrinate (1, 56, 61). Takatani et al. performed an experimental model documenting the selective role of this drug in the medial vestibular nucleus, which is fundamental for its function as a vestibular sedative. In terms of pharmacokinetics, it is considered a substrate and inhibitor of CYP2D6 (62), and it is also metabolized with minor affinity by CYP1A2, CYP2C9 and CYP2C19, properties that make it a drug with multiple interactions. In this regard, possible interactions with antihypertensives, central nervous system depressants, nonsteroidal anti-inflammatory drugs, and non-sedating vestibular antihistamines were identified in this study (56).
As for meclizine, a possible interaction with metoclopramide was observed. However, since it is the least prescribed active ingredient, the interaction occurred in a smaller proportion of cases. Meclizine is known to be an H1 receptor antagonist, which is metabolized by CYP2D6 and reduces the expression of CYP2B10, 3A11 and 1A2, thus explaining the multiple interactions with this drug (56).
No previous studies in Colombia have assessed the prevalence of the disease or the prescription patterns of vestibular sedatives. For that reason, this study provides an important background for the country's pharmacovigilance systems and may serve as a guideline for future research that contributes to the characterization of peripheral vertigo in the country.
There are limitations to this study. First, the data was obtained from the database of a logistic drug dispenser, so conclusions may only be drawn about groups with similar epidemiologic characteristics. Furthermore, the interpretation of some results was challenging since the records did not include the patients' clinical characteristics, disease severity, or adverse events that occurred throughout the research period.
Conclusions
Patients diagnosed with peripheral vertigo enrolled in the SGSSS who receive treatment with vestibular sedatives are mainly given betahistine and dimenhydrinate, antihistamines indicated for the management of this condition. Dimenhydrinate, accounting for 72.30%, is the most prescribed drug in the population studied. It is worth mentioning its use in patients diagnosed with benign paroxysmal vertigo, in whom repositioning maneuvers are recommended.
Based on the study conducted, it is evident that general practitioners, who are more involved in the prescription of vestibular sedatives, should receive more training in the treatment of peripheral vertigo, so that they can reach a more accurate diagnosis and establish an appropriate treatment based on each etiology.
Most patients received treatment for less than 3 months, which is consistent with the acute stage of the disease. However, it was found that betahistine was the active ingredient with the longest prolonged use.
On the other hand, among the possible clinical risk interactions evaluated in the present study, the most frequently reported interaction was dimenhydrinate-hyoscine bromide, which exacerbates adverse effects due to their anticholinergic properties. Likewise, additive synergism was found in 4.80% of the study population, with 4.7% of them showing at least one additive synergism.
Finally, a high degree of polypharmacy was observed, even though the majority of the population is below 60 years of age. Consequently, it is recommended to carefully prescribe these medications based on the needs of each patient, while considering potential interactions and adverse reactions in order to ensure targeted and safe therapy.
To Dr. Jorge Enrique Machado, Dr. Luis Valladales, Juan Camilo Forero, and Alfredo Portilla, who provided their support to help us obtain the Audifarma database.
No funding was received for this study.
Conflicts of interest
There are no conflicts of interest arising from the conduct of this study.
Both authors contributed to the elaboration of the study protocol, statistical analysis, and drafting of the article for subsequent publication.
Ethical considerations
The present observational study complies with Article 11 in Resolution 008430 of October 4, 1993 (“Scientific, technical and administrative standard for health research”) issued by the Ministry of Health, classifying this study as a “Risk-free research”, given that it employs a retrospective research method in which no biological, physiological, psychological, or social variables of the individuals to be studied are going to be modified. It was approved by the Ethics Committee for Research involving Human Subjects (CEISH) of the Sociedad de Cirugía de Bogotá, Hospital de San José (FUCS).
References
1. Bhattacharyya N, Gubbels SP, Schwartz SR, Edlow JA, El-Kashlan H, Fife T, et al. Clinical practice guideline: Benign paroxysmal positional vertigo (update). Otolaryngol Head Neck Surg. 2017;156(3_suppl):S1–47. doi: 10.1177/0194599816689667
2. Davis, Adrian & Moorjani, Padma. (2003). The Epidemiology of Hearing and Balance Disorders. Textbook of Audiological Medicine. 10.1201/b14730-8.
3. Welgampola MS, Bradshaw AP, Halmagyi GM. Assessment of the Vestibular System: History and Physical Examination. Adv Otorhinolaryngol. 2019;82:1-11. doi: 10.1159/000490267
4. Neuhauser HK. The epidemiology of dizziness and vertigo. En: Handbook of Clinical Neurology. Elsevier; 2016. p. 67–82.
5. Muelleman T, Shew M, Subbarayan R, Shum A, Sykes K, Staecker H, et al. Epidemiology of dizzy patient population in a neurotology clinic and predictors of peripheral etiology. Otol Neurotol. 2017;38(6):870-5. doi: 10.1097/MAO.0000000000001429
6. Murphy C, Reinhardt C, Linehan D, Katiri R, O’Connor A. A review of primary care referrals for patients with dizziness and vertigo: prevalence and demographics. Ir J Med Sci. 2022;191(1):385-9. doi: 10.1007/s11845-021-02575-6
7. Harun A, Agrawal Y. The use of fall risk increasing drugs (FRIDs) in patients with dizziness presenting to a neurotology clinic. Otol Neurotol. 2015;36(5):862–4. doi: 10.1097/mao.0000000000000742
8. Kaufman DW, Kelly JP, Rosenberg L, Anderson TE, Mitchell AA. Recent patterns of medication use in the ambulatory adult population of the United States: the Slone survey. JAMA. 2002;287(3):337–44. doi: 10.1001/jama.287.3.337
9. Herings RM, Stricker BH, de Boer A, Bakker A, Sturmans F. Benzodiazepines and the risk of falling leading to femur fractures. Dosage more important than elimination half-life. Arch Intern Med. 1995;155(16):1801-7.
10. Tinetti ME, McAvay GJ, Fried TR, Allore HG, Salmon JC, Foody JM, et al. Health outcome priorities among competing cardiovascular, fall injury, and medication-related symptom outcomes. J Am Geriatr Soc. 2008;56(8):1409-16. doi: 10.1111/j.1532-5415.2008.01815.x
11. Kasbekar AV, Mullin N, Morrow C, Youssef AM, Kay T, Lesser TH. Development of a physiotherapy-led balance clinic: the Aintree model. J Laryngol Otol. 2014;128(11):966-71. doi: 10.1017/S0022215114002060
12. Gurley KL, Edlow JA. Acute Dizziness. Semin Neurol. 2019;39(1):27-40. doi: 10.1055/s-0038-1676857
13. Royal College of Physicians. Hearing and Balance Disorders: Achieving Excellence in Diagnosis and Management. Report of a Working Party. London (United Kingdom): Royal College of Physicians. 2007;iii-65.
14. Burrows L, Lesser TH, Kasbekar AV, Roland N, Billing M. Independent prescriber physiotherapist led balance clinic: the Southport and Ormskirk pathway. J Laryngol Otol. 2017;131(5):417-424. doi: 10.1017/S0022215117000342
15. Trinidade A, Yung MW. Consultant-led, multidisciplinary balance clinic: process evaluation of a specialist model of care in a district general hospital. Clin Otolaryngol. 2014;39(2):95-101. doi: 10.1111/coa.12236
16. WHO Collaborating Centre for Drug Statistics Methodology (Internet). ATC/DDD Index 2024. Who.int. [citado el 16 de mayo de 2023]. Disponible en: https://atcddd.fhi.no/atc_ddd_index/updates_included_in_the_atc_ddd_index/
17. The selection of essential drugs: report of a WHO expert committee. World Health Organ Tech Rep Ser. 1977;(615):1-36.
18. Lee D, Bergman U. Studies of drug utilization. In: BL Strom, SF Kimmel, S Hennessy (eds). Pharmacoepidemiology, 5.a edición. John Wiley, Chichester, UK, 2012; 379–401.
19. Bergamasco A, Arredondo Bisono T, Castillon G, Moride Y. Drug Utilization Studies in Latin America: A Scoping Review and Survey of Ethical Requirements. Value Health Reg Issues. 2018;17:189-193. doi: 10.1016/j.vhri.2018.09.003
20. Masnoon N, Shakib S, Kalisch-Ellett L, Caughey GE. What is polypharmacy? A systematic review of definitions. BMC Geriatr. 2017;17(1):230. doi: 10.1186/s12877-017-0621-2
21. Dyhrfjeld-Johnsen J, Attali P. Management of peripheral vertigo with antihistamines: New options on the horizon. Br J Clin Pharmacol. 2019;85(10):2255-2263. doi: 10.1111/bcp.14046
22. Iwasaki S, Yamasoba T. Dizziness and Imbalance in the Elderly: Age-related Decline in the Vestibular System. Aging Dis. 2014;6(1):38-47. doi: 10.14336/AD.2014.0128
23. Lin E, Aligene K. Pharmacology of balance and dizziness. NeuroRehabilitation. 2013;32(3):529-42. doi: 10.3233/NRE-130875
24. Spiegel R, Rust H, Baumann T, Friedrich H, Sutter R, Göldlin M, et al. Treatment of dizziness: an interdisciplinary update. Swiss Med Wkly. 2017;147:w14566. doi: 10.4414/smw.2017.14566
25. Dunlap PM, Khoja SS, Whitney SL, Freburger JK. Assessment of Health Care Utilization for Dizziness in Ambulatory Care Settings in the United States. Otol Neurotol. 2019;40(9):e918-e924. doi: 10.1097/MAO.0000000000002359
26. Lee C, Jones TA. Neuropharmacological Targets for Drug Action in Vestibular Sensory Pathways. J Audiol Otol. 2017;21(3):125-132. doi: 10.7874/jao.2017.00171
27. Soto E, Vega R, Seseña E. Neuropharmacological basis of vestibular system disorder treatment. J Vestib Res. 2013;23(3):119-37. doi: 10.3233/VES-130494
28. Casani AP, Gufoni M, Capobianco S. Current Insights into Treating Vertigo in Older Adults. Drugs Aging. 2021;38(8):655-670. doi: 10.1007/s40266-021-00877-z
29. Gurumukhani JK, Patel DM, Shah SV, Patel MV, Patel MM, Patel AV. Negative Impact of Vestibular Suppressant Drugs on Provocative Positional Tests of BPPV: A Study from the Western Part of India. Ann Indian Acad Neurol. 2021;24(3):367-371. doi: 10.4103/aian.AIAN_413_20
30. Hülse R, Biesdorf A, Hörmann K, Stuck B, Erhart M, Hülse M, et al. Peripheral Vestibular Disorders: An Epidemiologic Survey in 70 Million Individuals. Otol Neurotol. 2019;40(1):88-95. doi: 10.1097/MAO.0000000000002013
31. Hunter BR, Wang AZ, Bucca AW, Musey PI Jr, Strachan CC, Roumpf SK, et al. Efficacy of Benzodiazepines or Antihistamines for Patients With Acute Vertigo: A Systematic Review and Meta-analysis. JAMA Neurol. 2022;79(9):846-855. doi: 10.1001/jamaneurol.2022.1858
32. Zwergal A, Strupp M, Brandt T. Advances in pharmacotherapy of vestibular and ocular motor disorders. Expert Opin Pharmacother. 2019;20(10):1267-1276. doi: 10.1080/14656566.2019.1610386
33. Michel L, Laurent T, Alain T. Rehabilitation of dynamic visual acuity in patients with unilateral vestibular hypofunction: earlier is better. Eur Arch Otorhinolaryngol. 2020;277(1):103-113. doi: 10.1007/s00405-019-05690-4
34. Schappert SM. National Ambulatory Medical Care Survey: 1989 summary. Vital Health Stat 13. 1992;(110):1-80.
35. Hanley K, O'Dowd T, Considine N. A systematic review of vertigo in primary care. Br J Gen Pract. 2001 Aug;51(469):666-71.
36. Nsson R J, Sixt E, Landahl S, Rosenhall U. Prevalence of dizziness and vertigo in an urban elderly population. J Vestib Res. 2004;14:47–52.
37. Furman JM, Raz Y, Whitney SL. Geriatric vestibulopathy assessment and management. Curr Opin Otolaryngol Head Neck Surg. 2010;18(5):386-91. doi: 10.1097/MOO.0b013e32833ce5a6
38. Ribeiro KF, Oliveira BS, Freitas RV, Ferreira LM, Deshpande N, Guerra RO. Effectiveness of Otolith Repositioning Maneuvers and Vestibular Rehabilitation exercises in elderly people with Benign Paroxysmal Positional Vertigo: a systematic review. Braz J Otorhinolaryngol. 2017;84(1):109–18. doi: 10.1016/j.bjorl.2017.06.003
39. Vaduva C, Estéban-Sánchez J, Sanz-Fernández R, Martín-Sanz E. Prevalence and management of post-BPPV residual symptoms. Eur Arch Otorhinolaryngol. 2018;275(6):1429-1437. doi: 10.1007/s00405-018-4980-x
40. Lynn S, Pool A, Rose D, Brey R, Suman V. Randomized trial of the canalith repositioning procedure. Otolaryngol Head Neck Surg. 1995;113(6):712-20. doi: 10.1016/S0194-59989570010-2
41. Burton MJ, Eby TL, Rosenfeld RM. Extracts from the Cochrane Library: modifications of the Epley (canalith repositioning) maneuver for posterior canal benign paroxysmal positional vertigo. Otolaryngol Head Neck Surg. 2012;147(3):407-11. doi: 10.1177/0194599812457134
42. Hain TC, Uddin M. Pharmacological treatment of vertigo. CNS Drugs. 2003;17(2):85-100. doi: 10.2165/00023210-200317020-00002
43. Shih RD, Walsh B, Eskin B, Allegra J, Fiesseler FW, Salo D, et al. Diazepam and Meclizine Are Equally Effective in the Treatment of Vertigo: An Emergency Department Randomized Double-Blind Placebo-Controlled Trial. J Emerg Med. 2017;52(1):23-27. doi: 10.1016/j.jemermed.2016.09.016
44. Lacour M, van de Heyning PH, Novotny M, Tighilet B. Betahistine in the treatment of Ménière's disease. Neuropsychiatr Dis Treat. 2007;3(4):429-40.
45. Tighilet B, Léonard J, Watabe I, Bernard-Demanze L, Lacour M. Betahistine Treatment in a Cat Model of Vestibular Pathology: Pharmacokinetic and Pharmacodynamic Approaches. Front Neurol. 2018;9:431. doi: 10.3389/fneur.2018.00431
46. Yu L, Zhang X-Y, Cao S-L, Peng S-Y, Ji D-Y, Zhu J-N, et al. Na(+) -Ca(2+) exchanger, leak K(+) channel and hyperpolarization-activated cyclic nucleotide-gated channel comediate the histamine-induced excitation on rat inferior vestibular nucleus neurons. CNS Neurosci Ther. 2016;22(3):184–93. doi: 10.1111/cns.12451
47. Haas H, Panula P. The role of histamine and the tuberomamillary nucleus in the nervous system. Nat Rev Neurosci. 2003;4(2):121-30. doi: 10.1038/nrn1034
48. Botta L, Mira E, Valli S, Zucca G, Benvenuti C, Fossati A, et al. Effects of betahistine and of its metabolites on vestibular sensory organs. Acta Otorhinolaryngol Ital. 2001;21(3 Suppl 66):24-30.
49. Murdin L, Hussain K, Schilder AG. Betahistine for symptoms of vertigo. Cochrane Database Syst Rev. 2016;2016(6):CD010696. doi: 10.1002/14651858.CD010696.pub2
50. Casani AP, Navari E, Guidetti G, Lacour M. Good Clinical Approach: Delphi Consensus for the Use of Betahistine in Menière's Disease. Int J Otolaryngol. 2018;2018:5359208. doi: 10.1155/2018/5359208
51. James AL, Burton MJ. Betahistine for Menière's disease or syndrome. Cochrane Database Syst Rev. 2001;2001(1):CD001873. doi: 10.1002/14651858.CD001873
52. Adrion C, Fischer CS, Wagner J, Gürkov R, Mansmann U, Strupp M; BEMED Study Group. Efficacy and safety of betahistine treatment in patients with Meniere's disease: primary results of a long term, multicentre, double blind, randomised, placebo controlled, dose defining trial (BEMED trial). BMJ. 2016;352:h6816. doi: 10.1136/bmj.h6816
53. Nauta JJ. Meta-analysis of clinical studies with betahistine in Ménière's disease and vestibular vertigo. Eur Arch Otorhinolaryngol. 2014;271(5):887-97. doi: 10.1007/s00405-013-2596-8
54. Della Pepa C, Guidetti G, Eandi M. Betahistine in the treatment of vertiginous syndromes: a meta-analysis. Acta Otorhinolaryngol Ital. 2006;26(4):208-15.
55. Strupp M, Kraus L, Schautzer F, Rujescu D. Menière's disease: combined pharmacotherapy with betahistine and the MAO-B inhibitor selegiline-an observational study. J Neurol. 2018;265(Suppl 1):80-85. doi: 10.1007/s00415-018-8809-8. Retraction in: J Neurol. 2020;267(4):1225. doi: 10.1007/s00415-020-09791-7
56. Di Mizio G, Marcianò G, Palleria C, Muraca L, Rania V, Roberti R, et al. Drug-Drug Interactions in Vestibular Diseases, Clinical Problems, and Medico-Legal Implications. Int J Environ Res Public Health. 2021;18(24):12936. doi: 10.3390/ijerph182412936
57. Gutiérrez-Valencia M, Izquierdo M, Cesari M, Casas-Herrero Á, Inzitari M, Martínez-Velilla N. The relationship between frailty and polypharmacy in older people: A systematic review. Br J Clin Pharmacol. 2018;84(7):1432-1444. doi: 10.1111/bcp.13590
58. Pit SW, Byles JE, Henry DA, Holt L, Hansen V, Bowman DA. A Quality Use of Medicines program for general practitioners and older people: a cluster randomised controlled trial. Med J Aust. 2007;187(1):23-30. doi: 10.5694/j.1326-5377.2007.tb01110.x
59. Chimirri S, Aiello R, Mazzitello C, Mumoli L, Palleria C, Altomonte M, et al. Vertigo/dizziness as a Drugs' adverse reaction. J Pharmacol Pharmacother. 2013;4(Suppl 1):S104-9. doi: 10.4103/0976-500X.120969
60. Kim HA, Bisdorff A, Bronstein AM, Lempert T, Rossi-Izquierdo M, Staab JP, et al. Hemodynamic orthostatic dizziness/vertigo: Diagnostic criteria. J Vestib Res. 2019;29(2-3):45-56. doi: 10.3233/VES-190655
61. Basura GJ, Adams ME, Monfared A, Schwartz SR, Antonelli PJ, Burkard R, et al. Clinical Practice Guideline: Ménière's Disease. Otolaryngol Head Neck Surg. 2020;162(2_suppl):S1-S55. doi: 10.1177/0194599820909438
62. Akutsu T, Kobayashi K, Sakurada K, Ikegaya H, Furihata T, Chiba K. Identification of human cytochrome p450 isozymes involved in diphenhydramine N-demethylation. Drug Metab Dispos. 2007;35(1):72-8. doi: 10.1124/dmd.106.012088